In the race to protect society from infectious microbes, the bugs are
outrunning us. The need for new therapeutic agents is acute, given the
emergence of novel pathogens as well as old foes bearing heightened
antibiotic resistance.
Shelley Haydel, a researcher at Arizona State University's Biodesign
Institute has a new approach to developing effective, topical
antibacterial agents — one that draws on a naturally occurring substance
recognized since antiquity for its medicinal properties: clay.
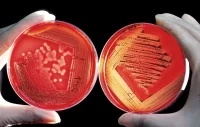
In research appearing in the journal PLOS ONE, Haydel and her
graduate student, Caitlin Otto, lay out the case for clay,
demonstrating that certain varieties of clay have the ability to
aggressively kill a range of pathogens including E. coli and methicillin-resistant Staphylococcus aureus
(MRSA) — a stubborn, highly contagious, and dangerous pathogen that has
lately been the scourge of many hospitals and is a common cause of skin
infections in the community. Their study further indicates that, rather
than the physical particles of the clays, particular metal ions
attached to the clay are likely responsible for its potent antibacterial
properties.
"While some natural clays, which have absorptive properties similar
to sponges, have been used topically for centuries, scientific studies
investigating the antibacterial mechanisms represent a relatively new
area of research," Haydel says. "With this study, we have demonstrated
that the antibacterial activity of these natural clays is not dependent
on the physical clay particles, but rather the abiotic, microbicidal
activities of specific metal ions desorbed from the clay surface. While
we are still working on mechanism of action studies, determining that
specific metal ions influenced antibacterial activity was critical in
leading us in the appropriate scientific directions."
Medical use of clay has a storied history. As early as 5000 years
ago, clay was listed in the ancient tablets of Nippur as a wound-healing
medicament. Around 1600 BC, the Ebers Papyrus—recognized as the
world's oldest medical text—recommended clay for ailments including
diarrhea, dysentery, tapeworm, hookworm, wounds, and abscesses. Clays
came into common use in the 19th century as topical treatments for
surgical wounds, demonstrating their beneficial effects for pain
management, inflammation, putrefaction, and healing processes.
In their current study, Otto and Haydel examined four clay samples
and their respective aqueous mineral extracts or leachates and
determined that the clays exhibited different in vitro antibacterial
activities against E. coli and MRSA. Mineralogically, the samples
were nearly identical with 52 percent clay and 48 percent non-clay
minerals, but the composition of metal ions released from the mineral
surfaces varied considerably across the samples. The tests, using
aqueous mineral leachates of the four clay samples, uncovered a variety
of elements in varying concentrations. Based on previous studies, the
research team focused on five metal ions—iron (Fe), copper (Cu), cobalt
(Co), nickel (Ni), and zinc (Zn).
When non-antibacterial clays with low concentrations of these five
critical metal ions were supplemented with higher amounts and the pH was
matched with that of antibacterial clays, the new formulation displayed
killing ability against E. coli and MRSA. The result pointed to
the presence of metal ions in sufficient concentration as the
antibacterial agent in the clay. Further tests narrowed the field of
antibacterial candidates, establishing Fe+2, Cu+2, and Zn+2 ions as
contributing antibacterial agents.
While the pH level was found to play a mediating role, the lethal
effect of the clays could not be attributed exclusively to pH, absent
the influence of metal ions. Metal speciation modeling and statistical
analysis of the results indicated that Cu+2, Co+2, Ni+2, and Zn+2 are
effective against E. coli, while Cu+2, Co+2, and Zn+2 are
effective against MRSA. Intriguingly, the study found that the metal ion
toxicity of a given clay sample is not always proportional to the total
ion concentration. Toxicity instead is critically dependent on a
variety of other factors including pH, ion solubility, osmotic strength,
and temperature. The tests undertaken helped to evaluate the interplay
of these factors in determining both the antibacterial effectiveness and
toxicity of the samples.
Haydel notes that physical and chemical properties of minerals
contained in clays together contribute to healing properties. Minerals
contained in clay mixtures have a negative surface charge that allows
the free exchange of compounds from the environment, including bacteria,
viruses, proteins, nucleic acids, and cations. Kaolinite, talc, and
smectite clay minerals are highly absorptive. Due to their ability to
adhere to the skin, clays offer mechanical protection similar to a
bandage, sealing out external physical or chemical agents, as well as
absorptive properties which assist in removing devitalized tissue,
particulate matter, or foreign materials from a wound.
Haydel is optimistic about the potential for medicinal clays to play
a greater therapeutic role, particularly against the growing threat of
topical and antibiotic-resistant infections:
"We have demonstrated that mineralogically-identical clays exhibit chemical variability which correlates with variability in antibacterial activity. Since clays can contain toxic metals, such as arsenic, cadmium, lead, and mercury, safety precautions must be in place to minimize exposure to toxic ions. Efforts must be taken to standardize the composition and antibacterial efficacy of clays if they are to be used therapeutically and prophylactically."
PLOS ONE paper: http://dx.plos.org/10.1371/journal.pone.0064068
Source: EurekAlert!